Compounded Peptides vs FDA-Approved Peptides: What's the Difference?
If you've been researching peptide therapy, you've probably noticed that some compounds are described as "FDA-approved" while others are called "compounded." The distinction matters more than most people realize. It affects the legal status of your treatment, the quality controls behind your medication, your provider's prescribing options, and your safety as a patient. Here's what you need to know.
What Does FDA-Approved Actually Mean?
An FDA-approved drug has gone through the agency's full review process: clinical trials demonstrating safety and efficacy in humans, manufacturing standards inspections, and post-market surveillance. Approval is granted for a specific compound, at a specific dose, for a specific indication. The manufacturer must meet Current Good Manufacturing Practice (cGMP) standards at every stage.
In the context of peptides, FDA-approved compounds include:
- Semaglutide (Ozempic, Wegovy) — approved for type 2 diabetes and chronic weight management
- Tirzepatide (Mounjaro, Zepbound) — approved for type 2 diabetes and obesity
- Tesamorelin (Egrifta) — approved for HIV-associated lipodystrophy
- Sermorelin — historically approved for growth hormone deficiency in children (approval withdrawn after market withdrawal, though clinical use continues)
- Liraglutide (Victoza, Saxenda) — approved for diabetes and weight management
FDA approval means there is a defined body of evidence supporting the drug's use for its labeled indication. It does not mean the drug is risk-free, nor does it mean it works for every patient or every use case.
What Is a Compounded Peptide?
Compounding is the practice of preparing a customized medication for an individual patient. A licensed compounding pharmacy combines, mixes, or alters pharmaceutical ingredients to create a formulation that isn't commercially available, or that needs to be tailored to a patient's specific needs — a different dose, a different delivery method, or the removal of an ingredient the patient is allergic to.
Under Section 503A of the Federal Food, Drug, and Cosmetic Act, compounding pharmacies can legally prepare medications using bulk drug substances from the FDA's Category 1 list. This is the pathway through which most therapeutic peptides — BPC-157, ipamorelin, thymosin alpha-1, GHK-Cu, and others — are currently prescribed in the United States.
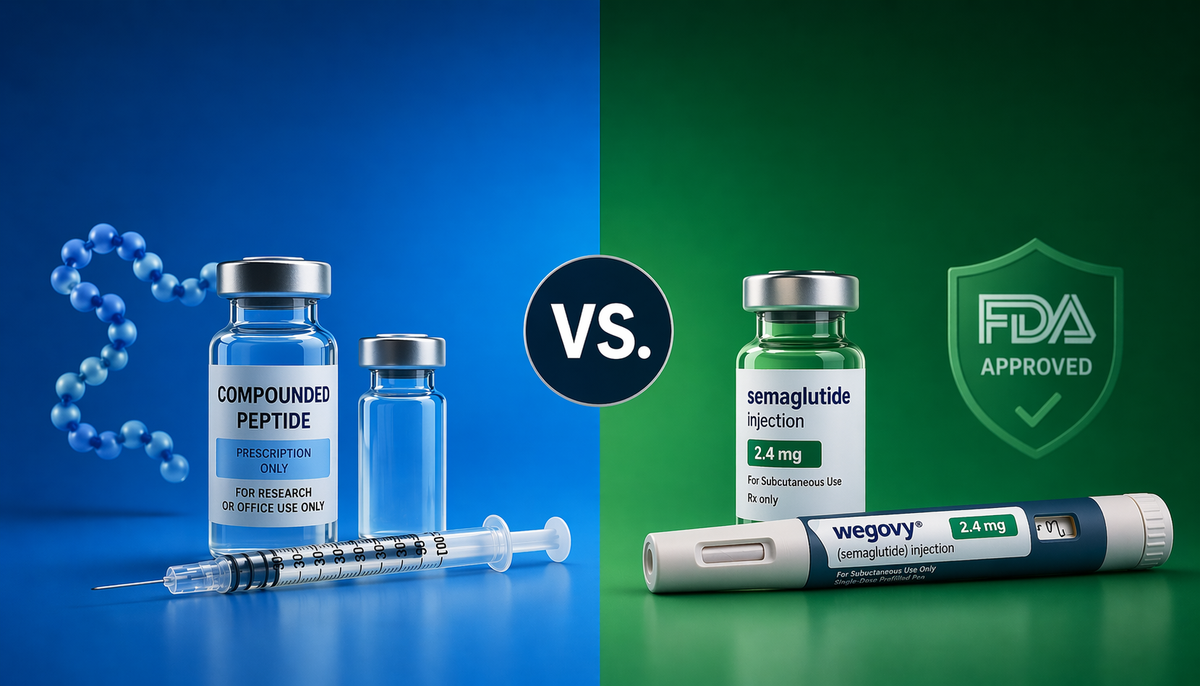
The key distinction: compounded peptides are not FDA-approved products. They have not gone through the agency's drug review process. But they are legal, regulated, and subject to quality standards when prepared by a licensed 503A pharmacy following USP guidelines.
Why Do So Many Peptides Exist Only as Compounded Products?
Most therapeutic peptides currently used in integrative and functional medicine have not gone through FDA drug approval for a simple reason: the approval process is extraordinarily expensive and time-consuming, often taking a decade and hundreds of millions of dollars. For peptides that cannot be patented — or that exist in a broad research and clinical interest space rather than a single defined disease indication — no pharmaceutical company has a commercial incentive to fund that process.
This does not mean the compounds are unproven or unsafe. It means the regulatory pathway has not been completed. The clinical and preclinical evidence base for compounds like BPC-157, thymosin alpha-1, and ipamorelin is substantial, even without FDA approval.
Compounding under 503A provides a legal framework for licensed providers to prescribe these compounds for individual patients when there is a clinical rationale to do so.
How Are They Different in Practice?
Quality control
FDA-approved drugs are manufactured in cGMP-certified facilities subject to regular FDA inspections. Every batch must meet strict specifications for purity, potency, and sterility before release.
Compounded medications prepared by licensed 503A pharmacies are subject to USP Chapter 797 standards for sterile preparations. These are rigorous requirements — but they are evaluated at the state level rather than by the FDA directly, and the oversight is less uniform than in pharmaceutical manufacturing.
Indication and labeling
FDA-approved drugs carry official labeling with a specific approved use. A compounded peptide is prescribed off-label, based on a provider's clinical judgment about an individual patient's needs.
Insurance coverage
FDA-approved drugs may be covered by insurance (subject to formulary and prior authorization). Compounded medications are almost never covered. This is a real practical consideration for patients.
Availability
FDA-approved peptides are manufactured at commercial scale and available through standard pharmacies. Compounded peptides require a prescription sent to a licensed compounding pharmacy, which then prepares the formulation for that specific patient.
What About the 2026 Reclassification?
In early 2026, the FDA moved 14 previously restricted peptides back to Category 1 under Section 503A, restoring their legal compounding status after a 2023 decision had cut off access. This reclassification did not make these compounds FDA-approved. It restored the legal pathway for licensed compounding pharmacies to prepare them as personalized prescriptions.
The distinction remains important: legal compounding access is not the same as FDA approval. Compounded peptides prescribed through a licensed clinical pathway are legal and subject to quality oversight, but they do not carry the same level of clinical evidence or regulatory review as approved drugs.
Which Is Better for You?
Neither category is universally superior. The right choice depends on what you're trying to address and what options exist.
For conditions where an FDA-approved peptide therapy exists and is appropriate for your situation, the approved product offers the most robust safety and efficacy data, is more likely to be manufactured to the highest quality standards, and may be covered by insurance.
For goals that don't match an approved indication — or for compounds that are only available through compounding — working with a licensed provider through a 503A pharmacy is the appropriate path. The key is choosing a licensed pharmacy, having a real clinical assessment, and not sourcing compounds outside the regulated system.
A licensed provider can help you understand which options apply to your situation and what the evidence supports for each.
Frequently Asked Questions
Are compounded peptides legal?
Yes, when prescribed by a licensed provider and prepared by a state-licensed 503A compounding pharmacy using Category 1 bulk substances. Compounded peptides sold without a prescription or by unlicensed vendors are not operating within this legal framework.
Are FDA-approved peptides safer than compounded ones?
FDA-approved drugs have more extensive clinical trial data and are manufactured under cGMP standards with direct FDA oversight. Licensed compounding pharmacies follow USP 797 standards, which are rigorous but involve less direct federal oversight. Both carry meaningful quality controls that gray-market products do not.
Can my doctor prescribe a compounded peptide?
Yes. Any licensed prescriber can write a prescription for a compounded medication. The prescription goes to a licensed 503A compounding pharmacy rather than a retail chain pharmacy.
Why isn't BPC-157 FDA-approved?
No pharmaceutical company has sought approval for BPC-157 as a drug, partly because it cannot be patented and no single company has a financial incentive to fund the approval process. This is a common situation for therapeutic compounds that predate the modern drug approval era or exist primarily within compounding and research contexts.
Does compounding mean the peptide is lower quality?
Not if it comes from a licensed, accredited pharmacy. The quality of compounded medications depends on the standards and practices of the pharmacy preparing them. Accredited 503A pharmacies operate under strict sterility, potency, and purity requirements. Gray-market products, regardless of how they are described, do not.
Sources
- U.S. Food & Drug Administration. Compounding and the FDA: Questions and Answers. FDA.gov
- Drug Quality and Security Act (DQSA), Pub. L. 113-54 (2013). Congress.gov
- USP General Chapter 797: Pharmaceutical Compounding — Sterile Preparations. USP.org
- U.S. Food & Drug Administration. FDA-Approved Drugs. FDA.gov
- McCall KL, et al. Safety analysis of compounded GLP-1 receptor agonists. *Expert Opinion on Drug Safety.* 2026;25(3):581-588.
This content is for educational purposes only and does not constitute medical advice. Peptide therapies should only be pursued under the supervision of a licensed healthcare provider. Amino Clinic recommends consulting with your physician before starting any new therapy.