The Difference Between Research Peptides and Pharmaceutical-Grade Peptides

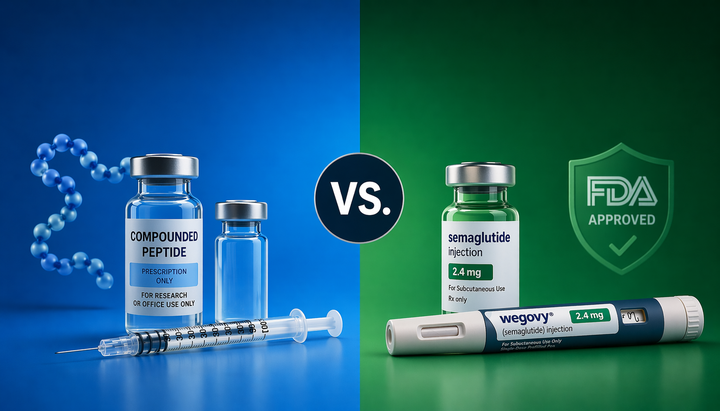
Walk through any corner of the internet where peptides are discussed and you'll find vendors selling compounds labeled "for research use only" or "not for human consumption." These are not pharmaceutical products. They are not the same as peptides prescribed by a licensed provider through a compounding pharmacy. Treating them as equivalent is a mistake that carries real health and legal consequences. Here's what the labels actually mean and why the distinction matters.
What Does "Research Use Only" Actually Mean?
"Research use only" (RUO) is a regulatory designation that means a product has not been evaluated for safety or efficacy in humans and is not intended for clinical use. In the United States, it is legal to manufacture and sell peptide compounds with this label without FDA oversight — provided they are genuinely sold for laboratory research purposes and not intended for human administration.
In practice, many vendors use the RUO label as a loophole. The compound is packaged, marketed, and sold in a format clearly intended for human use, with the RUO label attached primarily to avoid regulatory scrutiny. The FDA's March 2025 Final Guidance on research use only products made this distinction enforceable law: a product labeled RUO that is actually marketed for human use is in violation of FDA regulations, regardless of what the label says.
If you are buying a peptide from a website, injecting it yourself, and it came with no prescription and no provider involvement, you are using an RUO product — not a pharmaceutical-grade compound — regardless of how the vendor describes it.
What Is Pharmaceutical-Grade?
Pharmaceutical-grade refers to compounds that meet established pharmacopeial standards for identity, purity, potency, and quality. In the context of compounded peptides, pharmaceutical-grade means the bulk substance used by the compounding pharmacy meets USP (United States Pharmacopeia) or equivalent standards, and the finished preparation was made under USP Chapter 797 conditions for sterile compounding.
This involves:
- Verified identity of the active substance (it is what it says it is)
- Testing for purity and potency within specified ranges
- Sterility testing for injectable preparations
- Endotoxin testing to confirm absence of bacterial contaminants
- Preparation in a controlled environment by trained personnel
- Beyond-use dating that accounts for stability
None of this is required for RUO products. A vendor selling research peptides online has no obligation to test for purity, verify the compound's identity, test for sterility, or disclose what else is in the vial.
What the Independent Testing Shows
Several independent analyses have examined the quality of peptides sold through gray-market channels. The findings are consistent: a meaningful percentage of products contain the wrong compound, the wrong concentration, or contaminants not present in the labeled ingredient.
A 2024 study in Pharmaceutical Research examining follow-on and compounded GLP-1 peptide products (Hach et al.) found new impurities including high molecular weight proteins, trace metals, and residual solvents in products compared to originator formulations. Some oral products contained significantly less active compound than claimed on the label. This was in the context of semaglutide, which has enormous commercial interest and relatively strong supply chain oversight compared to less mainstream peptides.
For compounds like BPC-157 or ipamorelin, which have no commercial pharmaceutical manufacturer and are produced entirely by chemical synthesis operations with no FDA oversight, the quality variation is likely greater still.
The practical consequence of using a contaminated or mislabeled injectable compound is infection, allergic reaction, incorrect dosing with unknown effects, or in the worst cases, serious systemic harm from bacterial endotoxins or unidentified chemicals.
The Legal Dimension
Beyond the safety issues, there is a legal dimension that most consumers don't consider. Under U.S. federal law, introducing a drug into interstate commerce without FDA approval is illegal when that drug is intended for human use. Purchasing and self-administering an RUO peptide places you in a legal gray zone that is increasingly being scrutinized.
The FDA's March 2025 Final Guidance clarified the agency's position on RUO marketing: a product sold with RUO labeling but clearly marketed for human use is subject to enforcement action. Vendors caught in this space face legal consequences. The patients purchasing from them are in a murkier position, but one that is not entirely without risk — particularly as enforcement increases.
The licensed clinical pathway — prescription from a provider, dispensed by a licensed 503A pharmacy — is the only unambiguous legal route to human use of compounded peptides in the United States.
How to Tell the Difference
Practically speaking, here is how to identify whether a peptide is pharmaceutical-grade or a research compound:
Pharmaceutical-grade compounded peptide:
- Requires a prescription from a licensed healthcare provider
- Dispensed by a state-licensed compounding pharmacy
- Comes with a prescription label showing your name, the prescribing provider, directions, and pharmacy contact information
- Prepared under USP 797 sterile compounding standards
- Tested for identity, purity, potency, and sterility
Research use only peptide:
- No prescription required
- Ordered directly from a website or vendor
- May come in plain vials with minimal labeling
- Not subject to sterility or potency testing requirements
- No licensed provider involvement
The difference is not subtle. If you did not get a prescription from a licensed provider and pick up your peptide from a licensed pharmacy, it is not pharmaceutical-grade.
Why This Matters for Your Health Goals
People who pursue peptide therapy are typically doing so because they care about their health. They're interested in recovery, longevity, cognitive function, or managing a specific condition. The irony of sourcing compounds from unregulated vendors is that it directly undermines those goals.
A pharmaceutical-grade preparation gives you what the label claims at the concentration it claims, prepared under conditions designed to prevent contamination. An RUO product gives you an unknown. Even if the compound is the right one, the concentration may be wrong. Even if the concentration is right, there may be contaminants. And if you have an adverse reaction, there is no clinical oversight in place to catch it early or respond appropriately.
The legal pathway exists precisely because these compounds are biologically active and carry real risks when not used correctly. Working within it is not bureaucratic red tape. It is how you actually get what you're paying for.
Frequently Asked Questions
Are research peptides the same as compounded peptides?
No. Research use only peptides are not intended for human use under FDA regulations. Compounded peptides prescribed through a licensed provider and prepared by a licensed 503A pharmacy are legal pharmaceutical preparations subject to quality standards. They are fundamentally different products, regardless of what the underlying chemical compound is.
Is it illegal to buy research peptides for personal use?
Federal law prohibits introducing unapproved drugs intended for human use into interstate commerce. The FDA's 2025 Final Guidance reinforced this for RUO products marketed for human use. The legal risk sits primarily with vendors, but purchasing and self-administering these products is not without risk — and enforcement has increased.
How do I know if a compounding pharmacy is legitimate?
Look for a state pharmacy license (verifiable through your state's Board of Pharmacy) and PCAB (Pharmacy Compounding Accreditation Board) accreditation. A legitimate compounding pharmacy will provide its license information and be willing to answer questions about its sterility testing and quality assurance practices.
Why do people still buy research peptides if the risks are known?
Access and cost are the most common reasons. Until the 2026 reclassification, many peptides were not legally available through any compounding pathway, leaving gray-market sources as the only option for patients who wanted access. Cost is also a factor — RUO products are often cheaper than pharmacy-compounded versions. Neither rationale addresses the quality and safety gap.
Can a licensed provider help me transition from gray-market to clinical peptide use?
Yes. If you have been using research peptides and want to transition to a licensed clinical pathway, a provider can assess your current protocol, discuss what is now legally available through compounding, and develop a monitored treatment plan.
Sources
- U.S. Food & Drug Administration. Research Use Only Products: Final Guidance. March 2025. FDA.gov
- Hach M, et al. Impact of Manufacturing Process and Compounding on Properties and Quality of Follow-On GLP-1 Polypeptide Drugs. *Pharmaceutical Research.* 2024;41(10):1991-2014.
- McCall KL, et al. Safety analysis of compounded GLP-1 receptor agonists. *Expert Opinion on Drug Safety.* 2026;25(3):581-588.
- USP General Chapter 797: Pharmaceutical Compounding — Sterile Preparations. USP.org
- Drug Quality and Security Act (DQSA), Pub. L. 113-54 (2013). Congress.gov
This content is for educational purposes only and does not constitute medical advice. Peptide therapies should only be pursued under the supervision of a licensed healthcare provider. Amino Clinic recommends consulting with your physician before starting any new therapy.